EN
EN 55011: Industrial, scientific and medical equipment - Radio-frequency disturbance characteristics
Compliance Standards | EMC, Safety & Environmental Testing | ATEC
> EN Standards
> EN 55011: Industrial, scientific and medical equipment - Radio-frequency disturbance characteristics
EN 55011 Standard Overview
EN 55011 is a product family standard (largely based on CISPR 11) for industrial, scientific and medical (ISM) equipment and electro-discharge machining (EDM) and arc welding equipment. This standard sets forth procedures for the measurement of radio-frequency (RF) disturbances and limits within the frequency range of 9 kHz to 400 GHz. Requirements for ISM lighting apparatus and UV irradiators operating at frequencies within the ISM frequency bands defined by the ITU Radio Regulations are also contained in EN 55011.

ISM equipment or appliances tested against EN 55011 under the EMC Directive are designed to generate and use locally, RF energy for industrial, scientific, medical, domestic or similar purposes, excluding applications in the field of telecommunication. Typical ISM applications are the production of physical, biological, or chemical effects such as heating, ionization of gases, mechanical vibrations, hair removal and acceleration of charged particles.
Compliance with this standard gives a partial presumption of conformity with the European EMC Directive, 2004/108/EC. EN 55011 is an emission only standard covering radiated and conducted emissions. Most products will also require an assessment of immunity standards, such as:
(1) EN 61326-1 – Electrical equipment for measurement, control, and laboratory use; or
(2) EN 61000-6-2 – Generic Standards Immunity for industrial environments. These aforementioned two specifications contain additional requirements for electromagnetic interference and transient phenomenon. Additionally, devices powered via AC mains may require additional testing for power line harmonics and power line flicker.
Although the term medical is in the title of this standard, EN 55011 does not apply directly to medical devices within the scope of the Medical Device Directive 93/42/EEC. The reason is that for medical devices falling within the scope of 93/42/EEC, the EMC directive does not apply directly, but rather the EMC directive’s essential requirements are incorporated within the Medical Device Directive. Other standards harmonized under the Medical Device Directive, namely EN 60601-1-2 call upon EN 55011 to assess the electromagnetic disturbance characteristics of medical devices.
The manufacturer and/or supplier of ISM equipment shall ensure that the user is informed about the class and group of the equipment, either by labeling or by the accompanying documentation. In both cases, the manufacturer/supplier shall explain the meaning of both the class and the group in the documentation accompanying the equipment.
EN 55011 separates equipment into two groups:
EN 55011 Group 1 contains all ISM equipment in which there is intentionally generated and/or used conductively coupled radio-frequency energy which is necessary for the internal functioning of the equipment itself.
EN 55011 Group 2 contains all ISM equipment in which radio-frequency energy is intentionally generated and/or used in the form of electromagnetic radiation for the treatment of material, and EDM and arc welding equipment. Excluded from the testing requirements and limits of EN 55011 are components and subassemblies not intended to perform any stand-alone ISM function.
EN 55011 separates equipment of this type into two classes:
EN 55011 Class A is equipment suitable for use in all establishments other than domestic and those directly connected to a low voltage power supply network that supplies buildings used for domestic purposes.
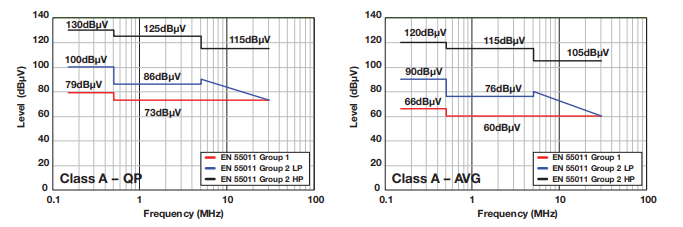
Class A Emission Limits
EN 55011 Class B is equipment suitable for use in domestic establishments and in establishments directly connected to a low voltage power supply network that supplies buildings used for domestic purposes.
ATEC offers affordable rental rates for signal analysis equipment from leading manufacturers like Tektronix, Keysight and Rohde & Schwarz.
Buy Standard

ISM equipment or appliances tested against EN 55011 under the EMC Directive are designed to generate and use locally, RF energy for industrial, scientific, medical, domestic or similar purposes, excluding applications in the field of telecommunication. Typical ISM applications are the production of physical, biological, or chemical effects such as heating, ionization of gases, mechanical vibrations, hair removal and acceleration of charged particles.
Compliance with this standard gives a partial presumption of conformity with the European EMC Directive, 2004/108/EC. EN 55011 is an emission only standard covering radiated and conducted emissions. Most products will also require an assessment of immunity standards, such as:
(1) EN 61326-1 – Electrical equipment for measurement, control, and laboratory use; or
(2) EN 61000-6-2 – Generic Standards Immunity for industrial environments. These aforementioned two specifications contain additional requirements for electromagnetic interference and transient phenomenon. Additionally, devices powered via AC mains may require additional testing for power line harmonics and power line flicker.
Although the term medical is in the title of this standard, EN 55011 does not apply directly to medical devices within the scope of the Medical Device Directive 93/42/EEC. The reason is that for medical devices falling within the scope of 93/42/EEC, the EMC directive does not apply directly, but rather the EMC directive’s essential requirements are incorporated within the Medical Device Directive. Other standards harmonized under the Medical Device Directive, namely EN 60601-1-2 call upon EN 55011 to assess the electromagnetic disturbance characteristics of medical devices.
The manufacturer and/or supplier of ISM equipment shall ensure that the user is informed about the class and group of the equipment, either by labeling or by the accompanying documentation. In both cases, the manufacturer/supplier shall explain the meaning of both the class and the group in the documentation accompanying the equipment.
EN 55011 separates equipment into two groups:
EN 55011 Group 1 contains all ISM equipment in which there is intentionally generated and/or used conductively coupled radio-frequency energy which is necessary for the internal functioning of the equipment itself.
EN 55011 Group 2 contains all ISM equipment in which radio-frequency energy is intentionally generated and/or used in the form of electromagnetic radiation for the treatment of material, and EDM and arc welding equipment. Excluded from the testing requirements and limits of EN 55011 are components and subassemblies not intended to perform any stand-alone ISM function.
EN 55011 separates equipment of this type into two classes:
EN 55011 Class A is equipment suitable for use in all establishments other than domestic and those directly connected to a low voltage power supply network that supplies buildings used for domestic purposes.
Class A Emission Limits

EN 55011 Class B is equipment suitable for use in domestic establishments and in establishments directly connected to a low voltage power supply network that supplies buildings used for domestic purposes.
ATEC offers affordable rental rates for signal analysis equipment from leading manufacturers like Tektronix, Keysight and Rohde & Schwarz.
Buy Standard
Products Used in Testing

Keysight 11947A Transient Limiter | 9 kHz – 200 MHz
- Impedance 50Ω
- Frequency range of 9 kHz - 200 MHz
- Insertion loss of 10 dB

PMM 9010 | 10 Hz – 30 MHz
- Frequency: 10Hz - 30MHz
- CISPR 16-1-1, MIL-STD-461 RE103
- Bandwidth: 200 Hz - 1 MHz (CISPR)


Rohde & Schwarz ESU40 | 20 Hz – 40 GHz
- Frequency: 10MHz/20Hz - 40 GHz
- CISPR 11, MIL-STD-461 RE103
- Bandwidth: Up to 30 MHz (FFT)


Rohde & Schwarz ESW44 EMI Receiver
- Frequency: 1 Hz - 44 GHz
- MIL-STD-461 CE106, Ford FMC1278
- 1 Hz - 10 MHz (80 MHz option)


Com-Power LI-125 LISN
- Current: 25A; Lines: 2
- Frequency: 150kHz - 30MHz
- Inductance: 50μH


Com-Power PC-114 Precompliance Test System
- Spectrum Analyzer
- Near Field Probes
- Biconical Antenna


Pacific Power 3150AFX AC & DC Programmable Power Source
- Frequency: DC, 15 – 1,200 Hz
- Accuracy: ± 0.01%
- 125 Arms / 62.5 ADC